An example of a simple production flow chart is proved below with a brief explanation of each step.
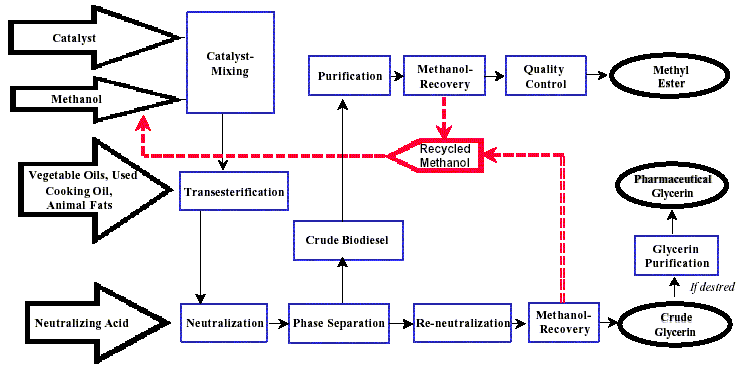
Mixing of alcohol and catalyst
The catalyst is typically sodium hydroxide (caustic soda) or potassium hydroxide (potash). It is dissolved in the alcohol using a standard agitator or mixer. Reaction. The alcohol/catalyst mix is then charged into a closed reaction vessel and the oil or fat is added. The system from here on is totally closed to the atmosphere to prevent the loss of alcohol. The reaction mix is kept just above the boiling point of the alcohol (around 160 °F) to speed up the reaction and the reaction takes place. Recommended reaction time varies from 1 to 8 hours, and some systems recommend the reaction take place at room temperature. Excess alcohol is normally used to ensure total conversion of the fat or oil to its esters. Care must be taken to monitor the amount of water and free fatty acids in the incoming oil or fat. If the free fatty acid level or water level is too high it may cause problems with soap formation and the separation of the glycerin by-product downstream.
Separation
Once the reaction is complete, two major products exist: glycerin and biodiesel. Each has a substantial amount of the excess methanol that was used in the reaction. The reacted mixture is sometimes neutralized at this step if needed. The glycerin phase is much more dense than biodiesel phase and the two can be gravity separated with glycerin simply drawn off the bottom of the settling vessel. In some cases, a centrifuge is used to separate the two materials faster.
Alcohol Removal
Once the glycerin and biodiesel phases have been separated, the excess alcohol in each phase is removed with a flash evaporation process or by distillation. In others systems, the alcohol is removed and the mixture neutralized before the glycerin and esters have been separated. In either case, the alcohol is recovered using distillation equipment and is re-used. Care must be taken to ensure no water accumulates in the recovered alcohol stream.
Glycerin Neutralization
The glycerin by-product contains unused catalyst and soaps that are neutralized with an acid and sent to storage as crude glycerin. In some cases the salt formed during this phase is recovered for use as fertilizer. In most cases the salt is left in the glycerin. Water and alcohol are removed to produce 80-88% pure glycerin that is ready to be sold as crude glycerin. In more sophisticated operations, the glycerin is distilled to 99% or higher purity and sold into the cosmetic and pharmaceutical markets.
Methyl Ester Wash
Once separated from the glycerin, the biodiesel is sometimes purified by washing gently with warm water to remove residual catalyst or soaps, dried, and sent to storage. In some processes this step is unnecessary. This is normally the end of the production process resulting in a clear amber-yellow liquid with a viscosity similar to petrodiesel. In some systems the biodiesel is distilled in an additional step to remove small amounts of color bodies to produce a colorless biodiesel.
Product Quality
Prior to use as a commercial fuel, the finished biodiesel must be analyzed using sophisticated analytical equipment to ensure it meets any required specifications. The most important aspects of biodiesel production to ensure trouble free operation in diesel engines are:
- Complete Reaction
- Removal of Glycerin
- Removal of Catalyst
- Removal of Alcohol
- Absence of Free Fatty Acids